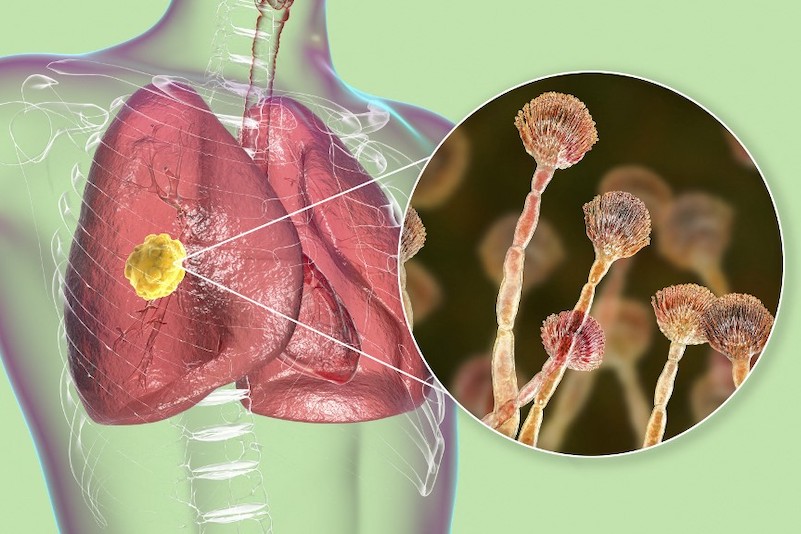
Japan’s Shionogi & Co has partnered with UK-based F2G to develop a drug for aspergillosis, a fungal infection that is spreading in Asia.
Olorofim is a novel oral therapy developed by F2G to treat invasive aspergillosis (IA) and other rare mould infections.
Shionogi will make an upfront payment to F2G of $100 million and share development costs in global studies. F2G will also be eligible to receive additional regulatory and commercial milestones of up to $380 million, as well as double-digit royalties on net sales.
IA is a rare disease that can be fatal, occurring primarily in immunocompromised patients, including those having received cancer chemotherapy or stem cell transplantation.
Olorofim is anticipated to be used to treat patients with serious invasive fungal disease where existing treatments are inappropriate or no longer effective. The success rate against IA in Asia has decreased due to increasing resistance, particularly in China.
Serious Infections
Both companies believe olorofim provides hope to patients with, and physicians treating, serious invasive fungal infections.
“In Asia, laboratory support in mycology in general, and for the diagnosis of IA in particular remains weak,” Tan Ban Hock, an infectious diseases specialist at Singapore General Hospital, said.
Treatment algorithms in Asia generally mirror those in the west, and the benefits of the new-generation azoles – antifungal compounds used to treat IA – have also been felt, he said.
“Access to these drugs, however, is a problem, with affordability a big issue among Asian clinicians,” Tan added.
“The majority of physicians managing an invasive fungal infection could not use the drug of choice because of cost,” said Tan, who is a member of the Asia Fungal Working Group, which studies the disease in China, India, Indonesia, Philippines, Singapore, Taiwan and Thailand.
- George Russell
READ MORE:
Shionogi & Co Shares Rise Over US Talks on Covid-19 Pill
Kintor’s Covid Drug Candidate Cuts Hospital, Death Risk
Super Fund Blasts Facebook as ‘Threat to Public Health’ – The Age